 References:ġ.”Isotopes of Hydrogen – Boundless Open Textbook.” Boundless. The main difference between Protium Deuterium and Tritium is that Protium has no neutrons in its nuclei while Deuterium is composed of one neutron and Tritium is composed of two neutrons. But they are highly unstable due to the presence of a high number of neutrons. Apart from these isotopes, there can be some other forms of hydrogen as well. Protium, Deuterium and Tritium are three isotopes of hydrogen. Tritium: The atomic mass of Tritium is 3.016 amu. Protium: The atomic mass of Protium is 1.00794 amu.ĭeuterium: The atomic mass of Deuterium is 2.014 amu. Tritium: The mass number of Tritium is 3. Protium: The mass number of Protium is 1.ĭeuterium:The mass number of Deuterium is 2. Chemical Symbolĭeuterium: The symbol for Deuterium is 1H. Tritium: Tritium is found in very trace amounts. Protium:The abundance of Protium is about 99.9%.ĭeuterium: The abundance of Deuterium is about 0.015%. Tritium:Tritium is an isotope of hydrogen that is composed of one proton, two neutrons, and one electron. Protium: Protium is an isotope of Hydrogen that is composed of one proton and one electron.ĭeuterium:Deuterium is an isotope of Hydrogen that is composed of one proton, one neutron, and one electron. However, the abundance of Tritium on earth’s crust is very less.įigure 4: Protium Deuterium Tritium Difference Between Protium Deuterium and Tritium Definition The half life of Tritium has been calculated as 12.32 years. 
This produces Helim-3 and it releases a large amount of energy. This isotope of hydrogen is radioactive due to the presence of a high number of neutrons compared to the number of protons. The atomic number of Tritium is 1 and the atomic mass of Tritium is 3. Tritium is an isotope of hydrogen that is composed of one proton, two neutrons, and one electron. Then, the peaks given by hydrogen atoms of the analyte can be distinguished by the atoms of the solvent. In NMR spectroscopy, Deuterium included compounds are used as the solvent instead of compounds composed of Hydrogen. There are some applications of Deuterium as well. Moreover, ice made from heavy water will sink in liquid water due to high density (normal ice floats on liquid water surface). Therefore, the bond length and bond energy are different from those of Protium. Due to the presence of the neutron, the atomic mass of Deuterium is as twice as Protium. Most of the times, Deuterium act in a similar manner as Protium. Heavy water is composed of D 2O molecules. If not, Deuterium can be found as heavy water. Deuterium exist as diatomic gases such as D 2 or HD (in combination with hydrogen). The occurrence of Deuterium can be either in gas phase or liquid phase. This is mainly because these atoms are minute and they have complete electron configurations in the only orbital (s orbital) in their diatomic molecule form. The bond between two atoms in the diatomic molecule has a higher bond dissociation enthalpy. Protium can be found in nature as a diatomic gaseous form or as hydrogen in H 2O molecule. The electron configuration of Protium is 1s 1. The atomic mass of Protium is about 1.00794 amu. The mass number of Protium is also 1 since there are no neutrons in the nucleus of Protium. The atomic number of Protium is 1 due to the presence of one proton. Therefore, when we are normally talking about hydrogen, we are talking about Protium. It is considered as the most stable isotope of hydrogen. The abundance of this isotope in the earth’s crust is about 99.9%. It is the most abundant form of hydrogen. Protium is an isotope of Hydrogen that is composed of one proton and one electron. Key Terms: Atomic Mass, Atomic Number, Deuterium, Isotopes, Neutron, Protium, Tritium What is Protium What is the Difference Between Protium Deuterium and Tritium What are the Similarities Between Protium Deuterium and Tritiumĥ. The main difference between Protium Deuterium and Tritium is that Protium has no neutrons in its nuclei whereas Deuterium is composed of one neutron and Tritium is composed of two neutrons. Therefore Protium, Deuterium, and Tritium share some similarities as well as differences. Due to this reason, isotopes have different physical properties but the chemical properties remain the same because the number of electrons present in isotopes is equal. Therefore, isotopes have the same atomic number but different atomic masses. 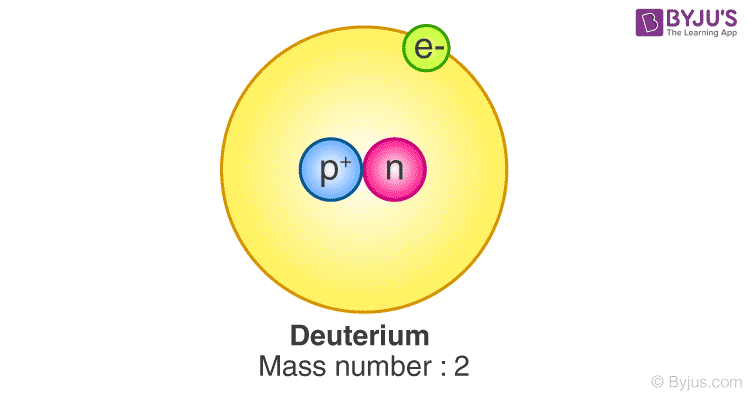
Isotopes are different forms of the same element that are different from each other according to the number of neutrons they have in their nuclei. 
Protium, Deuterium, and Tritium are isotopes of the Hydrogen element. 
Main Difference – Protium vs Deuterium vs Tritium 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
